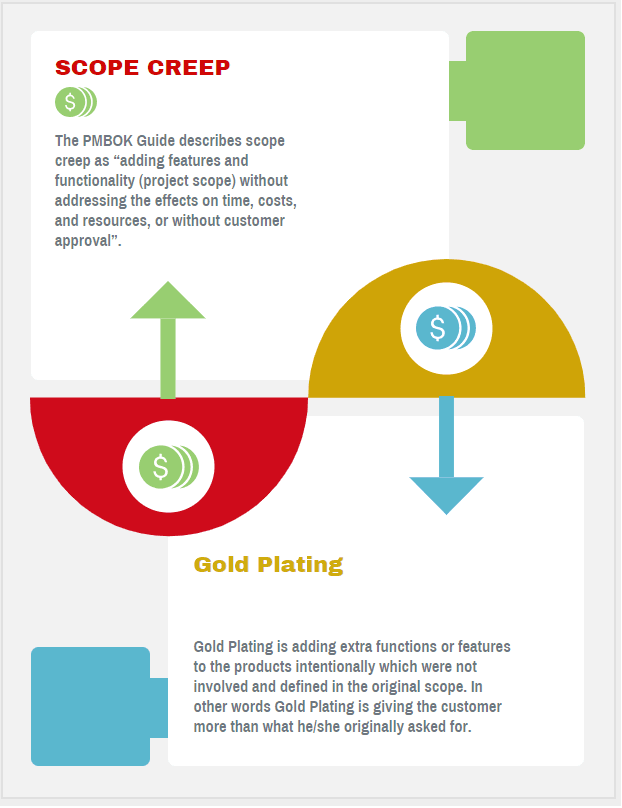
Although primitive, people from the early times have been using and designing groundbreaking pieces of technology to ease the load from certain tasks. In the medical field, technology is utilized the most to help with these tasks. Whether it’s by performing operations on patients or simply watching what it does, taking in how it works is already a pleasure. However, as time went by, the quality of this technology also skyrocketed. After all, you can already imagine the quality medical devices get once manufacturers work with the best medical device design company. Out of this quality, you’ve probably noticed these specialists pushing interactivity more in many of their designs. Here the interactive medical equipment takes the stage.
Table of Contents
Why Medical Equipment Needs Interactivity
Designing medical devices is a complicated task due to how much variety there is. As there are many specializations within the medical field, the diversity found from using specific medical equipment in each one is also there. However, they all have a universal purpose: to provide an optimal experience for the user and the patient.
With this in mind, designing most medical equipment to be interactive is a conscious decision. By making it interactive, it could accommodate its performance to what you’re looking for through customizations or by having an artificial intelligence draw out pre-calculated settings. To understand better, here are some common examples of interactive medical equipment:
- Defibrillators
- Vital sign monitors
- Heart pumps
As you can see, unlike scalpels or stethoscopes, these devices are designed to shoulder a variety of tasks during the operation, be it for diagnostics or other life-saving purposes. Thus, allowing some relief for the user and patient alike during and after a procedure.
Steps In Designing Interactive Medical Equipment
Most could agree that drawing out plans for anything tends to be more tedious than putting it into action. Yet when designing medical devices, being thorough is a necessity. Nevertheless, patient safety should always be highly regarded in medical settings. As for the users, ensuring their security in handling these devices promises a smoother performance.
1. Construct Ideas
This is the part where you and your team would brainstorm what features the device should have. However, before you could get started with that, ask yourself these questions first:
- Which specialization is your target?
- Is there a need for a device like yours in this specialization?
- Are you vying for some amount of uniqueness in your work?
Now, you might wonder why this resembles a business model too much. Remember that whatever design you make will go to healthcare providers when your manufacturers finish building them. Furthermore, by taking the time to review the market, your chances of hitting your target audience grow stronger each day.
2. Address Potential Risks
Interactive medical equipment is for the health workers to be more efficient in applying their practices to the patient. Broadly speaking, you can’t avoid risks when designing equipment. Although this might seem impractical (or ‘poorly made’), risks can arise regardless of how ‘top-of-the-line’ the manufacturer is.
What truly makes a medical device one of the best starts in acknowledging the potential dangers brought out while using it. For example, there’s no telling when a defibrillator could overload or cause a power outage or electrocution. Therefore, in laying out the design for the medical equipment, you must emphasize identifying the risks from every feature you’re planning to include.
Medical devices are vital instruments for modern healthcare delivery. However, major complications can develop if medical equipment are created for ‘work as imagined’ when this is mismatched with ‘work as done’. This challenge can be amplified since the nuances of device design, in terms of usability and the way a device supports or affects working behaviors, typically receives insufficient attention. The ways devices are built and utilized impact patient safety and quality of care. So, incorrect design can encourage user error, introduce system vulnerabilities and divert attention from other elements of patient care. Current regulation entails a number of pre-market inspections pertaining to device usability, but this implies that gadgets are always use under circumstances and for the reasons intended.
3. Install Risk Responses
Once you’ve identified the risks, design countermeasures that could help control the damage they’d cause. After all, as mentioned earlier, what makes a medical manufacturer approved for Medicare is knowing the risks their products could bring.
By acknowledging them for interactive medical equipment, only then could you construct safety measures if they ever come up. In doing so, the safety of both patient and user will be secure. However, it’s also why this phase is far more challenging than simply brainstorming for features. Not only are you need to point out the risks (be it predictable or otherwise) but your safety measures also need to remain 10 steps ahead of them.
4. Perform Experimental Runs
You could only predict how your design would fare by running it through multiple tests. Witnessing how it performs up close in a controlled setting can help you peruse through every possible situation where the problem could trigger. Whenever your design fails, consider it an opportunity to take note and be careful next time.
Takeaway
Devices are beginning to play significant part in the delivery of care, whether in monitoring vital signs, administration of drugs, analgesia or delivering specialty therapies. Technology’s gone a long way from only using manual medical devices for complex procedures. Because as time goes by, so does humanity’s drive to offer efficient solutions for everyone and it applies to interactive medical equipment as well. The integration of interactivity in medical equipment proves that.

Business Developmeny Manager at PAS InfoCom Technologies Ltd. Experienced in project management with a demonstrated history of working in the information technology and services industry.